Engineering for Health—Individual and Diverse
June 28, 2021
The production of drugs usually involves complex processes that are subject to extensive monitoring. For an Irish manufacturer of biopharmaceuticals, Pepperl+Fuchs has developed around thirty different individual solutions for its human machine interfaces (HMI), which employees can use to reliably control and monitor production processes.
Health plays a huge part in our quality of life, perhaps even the most important part. We have seen just how true this sentiment is during the COVID-19 pandemic with the restrictions that have been put in place and the pandemic’s impact on people and healthcare systems all over the world. Nevertheless, we sometimes still take our own health or that of our loved ones for granted. That is, until we get sick. Whether it’s flu, diabetes, rheumatic complaints, or even more serious illnesses, when you are directly affected, you appreciate what it means to be healthy, and you want to get back on your feet as soon as possible.
To make sure that we can recover, manufacturers of pharmaceutical products work continuously on drugs, some of which will save lives. Many billions of dollars are spent worldwide every year on research to find the right drug for a wide variety of diseases and to help those suffering quickly regain a better quality of life. Development of biopharmaceuticals is a growing trend that has emerged in the pharmaceutical industry during the last few years. This term describes pharmaceutical products produced using biotechnology. These products are based on micro-organisms or cell lines from mammals and plants, while conventional chemical pharmaceuticals are purely synthetic. Biotechnology is even spearheading the production of complex products that have an elaborate synthetic composition or one that cannot be manufactured. Biopharmaceutical products are usually produced in bioreactors or other closed containers. As with pharmaceutical production in general, certain regulations must also be observed in the field of biopharmaceuticals. The GMP (Good Manufacturing Practice) guidelines set extensive standards for quality, hygiene, cleanliness, and care, since contaminants and quality fluctuations in the pharmaceutical sector—whether chemical pharmaceuticals or biopharmaceuticals—can have fatal consequences.
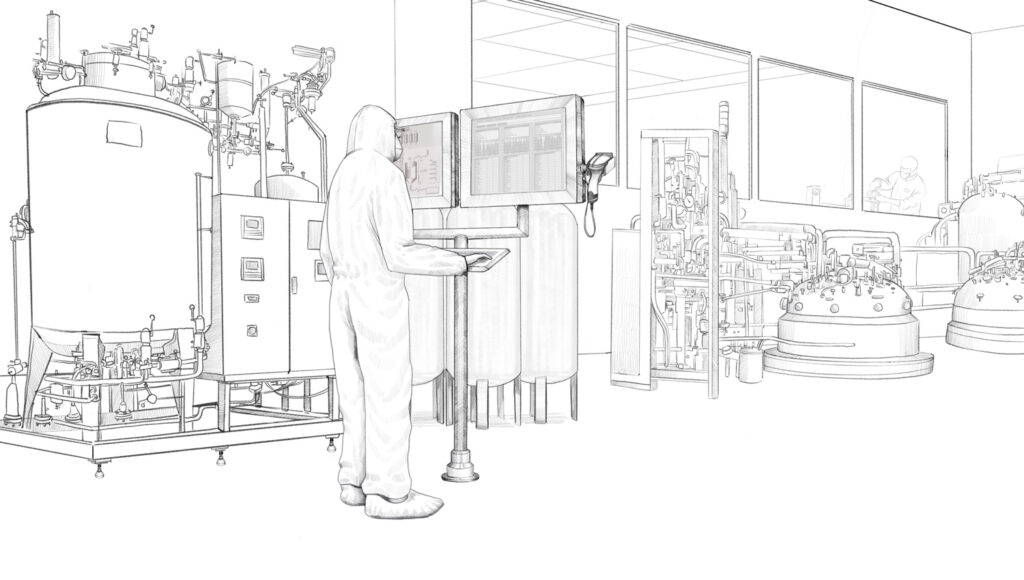
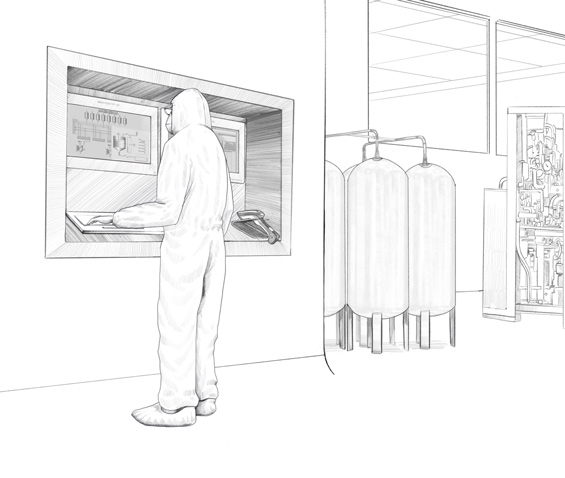
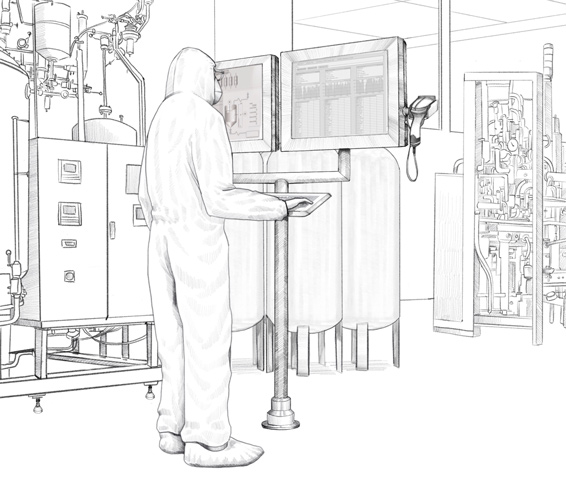
Customized HMI solution for production process monitoring in the manufacture of biopharmaceuticals.
Customized HMI solution for production process monitoring in the manufacture of biopharmaceuticals.
A Tailored Approach
To continually monitor the complex processes involved in the production of biopharmaceuticals, it is necessary to use products and technologies that allow all relevant information to be reliably accessed in the production area while also complying with strict GMP guidelines. For example, operating devices in the manufacturing areas must be insensitive to the chemical substances used there and easy to clean to ensure that bacteria or dirt cannot accumulate. With its portfolio of HMI systems, Pepperl+Fuchs offers a whole range of products that can be used in this environment. On top of that, our application experts around the world can advise biopharmaceuticals manufacturers in selecting the right components and, if desired, create a solution tailored to the manufacturer’s individual requirements and needs. Stefan Sittel, Business Development Manager HMI at Pepperl+Fuchs, is one such expert and understands the special requirements of customers in the biopharmaceutical sector: ‘The pharmaceutical industry is diverse, with a variety of factors to consider. Solutions need to be individually adjusted to accommodate the varied workplaces in production environments. Our engineered HMI solutions supplement our standard portfolio and allow us to meet these challenges.’

Especially in large-scale customer projects, a lot of engineering work is involved when implementing specific requirements because many different conditions within pharmaceutical production have to be considered. “We worked with a biopharmaceutical manufacturer in Ireland to perform extensive mechanical adjustments to around thirty industrial monitors for the non-hazardous area. Only two of the monitors were identical; all of the other monitors differed in some way,” says Sittel, recounting a project that was successfully completed. “You could say that we explored all possible adjustments in this project to adapt the monitors ideally to the conditions at the customer’s workplaces.”
Barely Two Alike
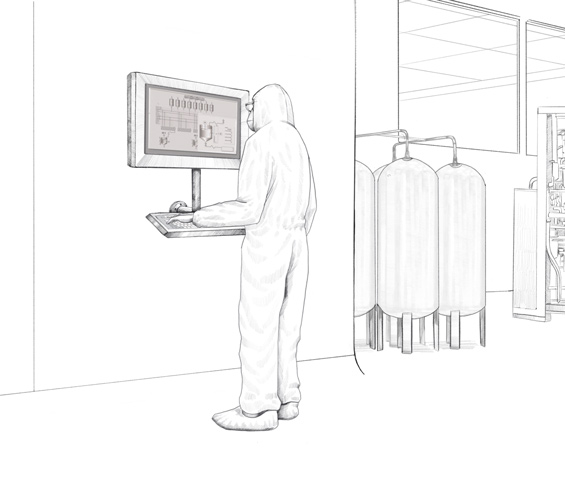
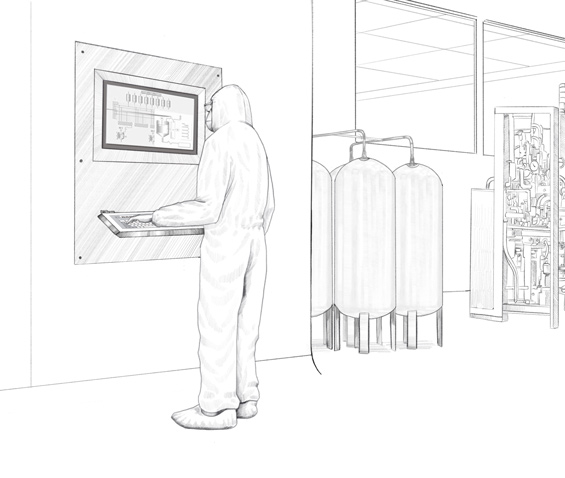
Remote and direct monitors from the VisuNet series from Pepperl+Fuchs are used in this biopharmaceutical plant. Specially developed for the requirements of process automation in the life science industry, these monitors are easy to clean and especially resistant to chemicals and cleaning agents. Pepperl+Fuchs individually developed and adapted the housings and mounting options for the Irish biopharmaceutical company so that the monitors could be adapted to the existing installation conditions on-site. In total, six different types of customized stainless steel housings were created. These housings can be mounted in different ways.
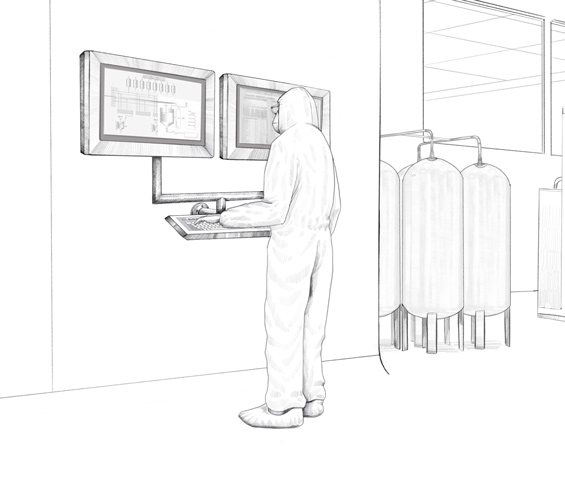
Working from one of the company’s own Solution Engineering Centers (SEC), the experts from Pepperl+Fuchs developed a monitor version to be fitted in different pharmaceutical walls plus multiple brackets for use with wall-mounted monitors. Both options—fitted in the wall and mounted on the wall—are available for both single and duplex monitors. The duplex designs allows the operator in the plant to simultaneously view different information on the two screens—the Manufacturing Execution System (MES), which controls production, and the Distributed Control System (DCS), i.e. the plant automation system. Pepperl+Fuchs manufactured various combinations of remote and direct monitors tailored to the customer’s requirements. Some duplex monitors were wall-mounted on-site while others were used in the plant as stand-alone versions on a pedestal.

To ensure that the peripheral devices are also ideally suited for the customer’s requirements, Pepperl+Fuchs replaced the keyboards that accompanied some monitors with a specially developed rack design. “Our customer wanted to use keyboards and mouses that had a hygienic design with a silicone coating instead of stainless steel keyboards. They wanted to make sure these items could be replaced easily if they became worn. Our custom-designed rack enables flexible usage of the accessories,” says Sittel, explaining the advantage of the individual design.
In the Best Hands
The individually adapted HMI solutions have been in use for around three years and in that time have been providing reliable support to those working in the plant to produce vital drugs. Personnel at the biopharmaceutical company always have access to all relevant information, while the VisuNet monitors ensure compliance with all regulations. Our customers, such as the Irish biopharmaceutical manufacturer, can take advantage of the engineered solutions offered by Pepperl+Fuchs to get the HMI system that is right for them.
“Our Solution Engineering Center benefits companies by putting the planning,engineering, and certification of customized HMI solutions solely in the hands of our experts, allowing the companies to concentrate on their own core business, such as producing drugs,” says Sittel. Application experts in the six SECs from Pepperl+Fuchs around the globe have the skills to adapt standard components from the HMI portfolio to fully meet specific customer requirements and provide companies with optimal support.
Watch video
Learn more
Our Solutions,
as Individual as You
Subscribe now
Would you like to find out more about customized solutions from Pepperl+Fuchs? Register via the form below to receive regular updates.
Receive regular updates on customized solutions from Pepperl+Fuchs!
Subscribe now